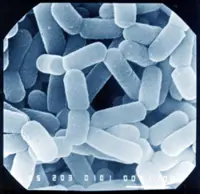
Before we learn how to kill bacteria, we should know what bacteria are and why we need to kill them. Bacteria are single-celled prokaryotic microorganisms found mostly under the Eubacteria kingdom in the Linnaeus classification.
A prokaryotic organism does not have a nucleus or membrane-bound organelles in its cells. In contrast, humans, animals and plants are all eukaryotic organisms that have nuclei and membrane-bound organelles in their cells.
Already a subscriber? Log in
Get 20% OFF The Star Digital Access
Cancel anytime. Ad-free. Unlimited access with perks.